 |
clinical documentation
|
clinical documentation
612 PROCEDURE GUIDE
Deep Melasma Protocol — Step-by-Step Clinical Application Guide for Practitioners
⚠ TCA-BASED PROTOCOL — ADVANCED PRACTITIONERS ONLY
prodermic 612 contains Trichloroacetic Acid (TCA). This is a medium-depth chemical peel that requires frost endpoint monitoring. It should only be performed by experienced dermatologists or trained aesthetic professionals with prior TCA peel experience. Improper application can result in scarring, post-inflammatory hyperpigmentation, or burns.
Product Overview

prodermic® 612
Deep Melasma Protocol · Cosmelan Forte
| Type | 7-Agent Combo Peel (Highest agent count) |
| Total Active | High concentration |
| Size | 30ml jar |
| Yield | ~7 sessions per jar |
| Fitzpatrick | II–IV (caution V, avoid VI) |
| Peel Depth | Superficial to Medium |
| Aggression | HIGH |
Active Composition
Mechanism of Action
| Agent | Mechanism |
|---|---|
| TCA | Controlled medium-depth peeling — trichloroacetic acid causes controlled coagulation necrosis of epidermis to access deep pigment deposits. The primary penetration driver of this protocol. |
| Arbutin | Hydroquinone glycoside (natural origin) — potent tyrosinase inhibitor that blocks melanin synthesis without the side effects associated with hydroquinone. |
| Niacinamide | Blocks melanosome transfer — prevents pigment from moving from melanocytes to surrounding keratinocytes. Also supports barrier repair. |
| Kojic Acid | Chelates copper required for tyrosinase enzymatic activity, providing a second independent pathway of pigment suppression. |
| Vitamin C | Antioxidant defense, collagen synthesis support, and mild tyrosinase inhibition for additional pigment control. |
| Glycolic & Lactic Acid | Multi-depth AHA surface resurfacing — glycolic provides rapid superficial exfoliation while lactic acid supports hydrating desquamation and ceramide production. |
Clinical Indications
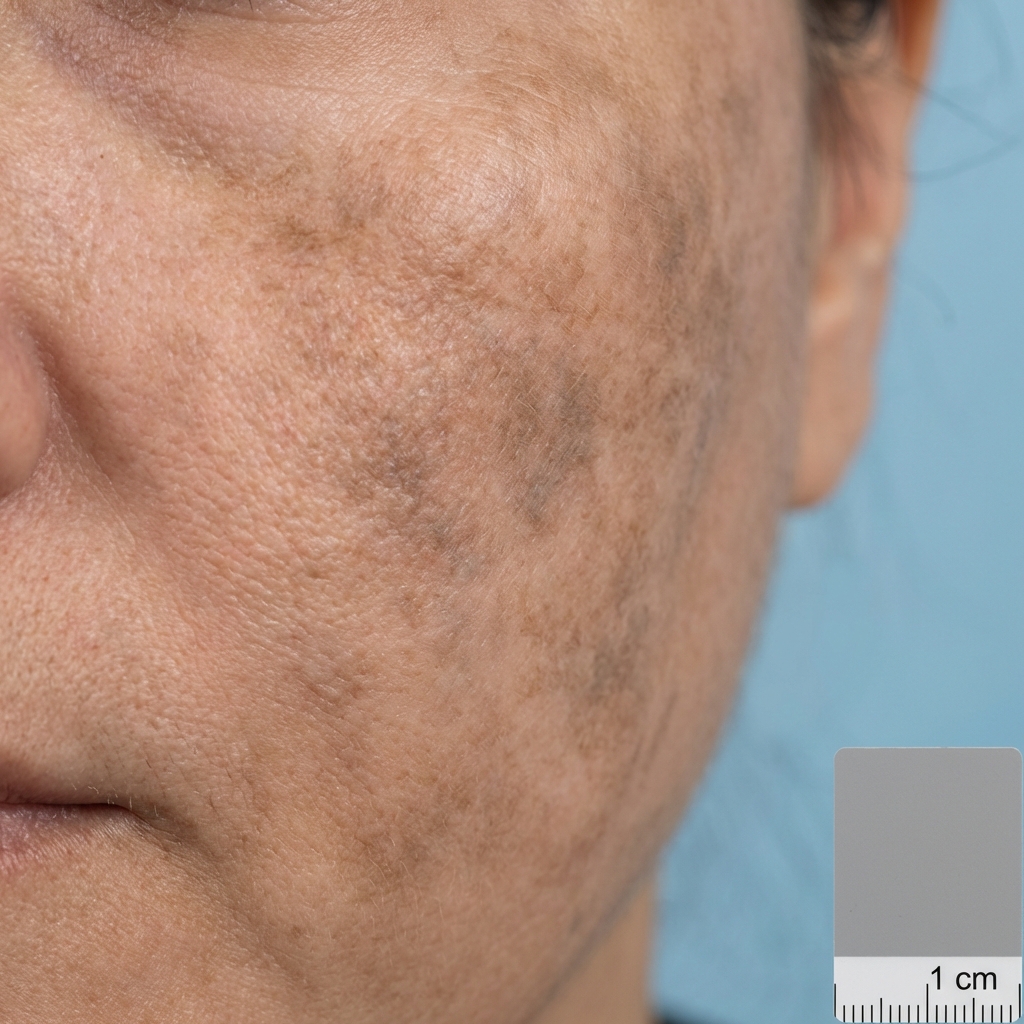
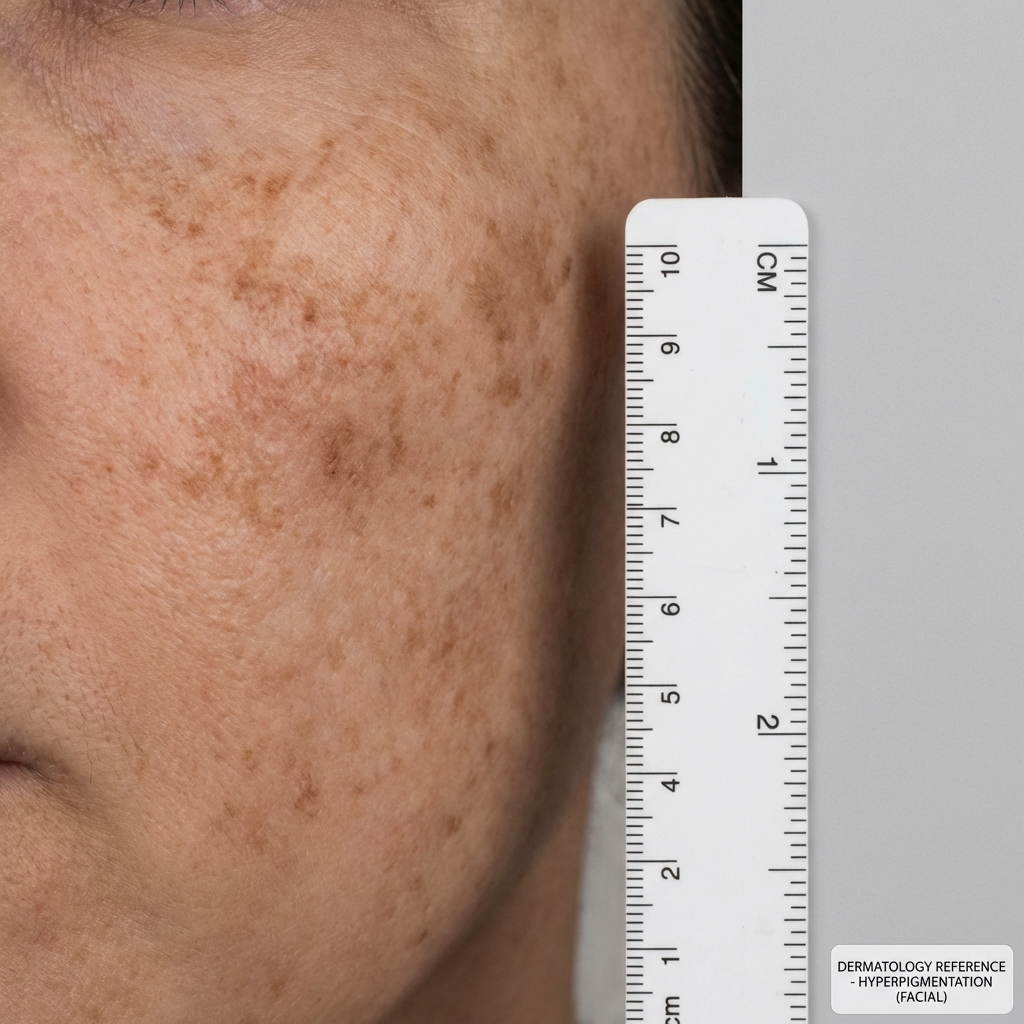
Patient Selection & Pre-Procedure
Ideal Patient Profile
- Treatment-resistant melasma that has not responded to standard AHA protocols
- Patients seeking a Cosmelan/Dermamelan alternative with comparable clinical outcomes
- Patients who are ready for a more aggressive intervention after milder peels have plateaued
- Dermal or mixed-type pigmentation requiring deeper penetration than superficial peels can achieve
- Fitzpatrick skin types II–IV (use with caution on type V — AVOID type VI)
- Patients who understand and accept the 5–10 day downtime commitment
⚠ FITZPATRICK VI — ABSOLUTE CONTRAINDICATION
- Do NOT use prodermic 612 on Fitzpatrick VI skin. The TCA component carries a high risk of post-inflammatory hyperpigmentation (PIH) and dyschromia in deeply pigmented skin. There is no safe way to titrate this risk in type VI patients.
- For Fitzpatrick V patients: proceed with extreme caution, conservative application, and reduced contact time. Consider a test area first and wait 4 weeks to assess for PIH before proceeding with full treatment.
Contraindications
DO NOT TREAT IF
- Active skin infections or inflammation in the treatment area
- Compromised skin barrier (eczema, dermatitis flare, rosacea flare)
- Pregnancy or breastfeeding
- Oral isotretinoin use within the last 6 months
- Topical retinoids use within 72 hours of session
- Open wounds, cuts, or abrasions in the treatment area
- Fitzpatrick VI skin (absolute contraindication)
- History of keloid or hypertrophic scarring
- History of post-inflammatory hyperpigmentation after previous chemical peels
- Active herpes simplex — TCA can trigger viral reactivation (consider antiviral prophylaxis for patients with HSV history)
- Known allergy to TCA or any of the active ingredients
- Recent laser or IPL treatment within 4 weeks
Pre-Procedure Patient Preparation
- Comprehensive Skin Assessment: Evaluate Fitzpatrick type, melasma pattern (epidermal vs. dermal vs. mixed — use Wood’s lamp), prior peel history, and any history of PIH. Determine suitability carefully.
- HSV Prophylaxis: For patients with a history of herpes simplex, prescribe antiviral prophylaxis (e.g., valacyclovir) starting 2 days before the procedure and continuing for 5 days after.
- Sun Exposure: Advise the patient to avoid all direct sun exposure for at least 2 weeks prior. Strict SPF 50+ use is mandatory in the lead-up.
- Discontinue: Stop topical retinoids 72 hours before. Stop all AHAs/BHAs 48 hours before. Stop any other chemical exfoliants.
- Informed Consent: Discuss expected outcomes, the frost monitoring process, significant downtime (5–10 days of peeling), the self-neutralizing nature (patient washes off at home), TCA-specific risks (PIH, scarring if misused), and the need for strict post-care compliance.
- Photography: Take standardized baseline photographs under consistent lighting. Include close-ups of pigmented areas.
Step-by-Step Procedure
KEY: SELF-NEUTRALIZING FORMULA WITH FROST ENDPOINT
prodermic 612 is a self-neutralizing peel with TCA. It is applied in-clinic with frost endpoint monitoring. Once the frost develops and stabilizes, the peel begins self-neutralizing. The patient leaves with the peel on and washes it off at home after 4–6 hours. No in-clinic neutralization agent is required.
- Forehead: Apply in horizontal strokes from center outward, even pressure
- Cheeks: Apply from the nose outward toward the ears — most common melasma zone
- Nose bridge: Light application only
- Chin: Apply in upward strokes
⚠ FROST MONITORING — CRITICAL CLINICAL STEP
Frost level determines peel depth. You must monitor the frost development continuously and know when to stop. The frost endpoint is the most important safety variable in this protocol.
- Frost stabilization then gradual fade: Normal — this is the expected response
- Continued frost deepening: Monitor closely; if progressing to solid white, be prepared for extended post-care
- Severe burning, swelling, or blister formation: Remove product immediately with saline-soaked gauze
- Leave the peel on: The patient leaves with the peel still on the skin
- Contact time: Instruct the patient to leave it on for 4–6 hours (based on session number — see Progressive Protocol)
- Wash off at home: After the prescribed contact time, rinse thoroughly with lukewarm water until all residue is completely removed
- Apply a thick, bland recovery moisturizer or barrier cream immediately after washing
- No other actives, serums, or makeup for the rest of the day
Early Removal & Adverse Reactions
INSTRUCT PATIENT TO WASH OFF IMMEDIATELY IF
- Severe burning or pain that does not subside or is escalating after the first hour
- Excessive redness, edema, or swelling beyond what was observed in-clinic
- Hives, welts, or signs of allergic reaction
- Blistering or skin breakdown (rare but possible with TCA)
- Inability to tolerate the sensation — patient safety and comfort come first
Emergency Removal Steps (At Home)
- Rinse the treatment area thoroughly with cool water (not cold — avoid thermal shock) until all product is removed
- Continue rinsing until all tingling and burning has completely stopped
- Apply a thick, bland barrier cream or petrolatum-based ointment (no actives)
- Apply a cool compress for comfort if needed
- Contact the clinic immediately to report the reaction — send photographs
- Do not apply any other products until instructed by the practitioner
- Document the reaction thoroughly — reduce contact time, application density, or consider stepping down to a milder protocol (e.g., 580) for the next session
PATIENT COMMUNICATION
- Ensure the patient has the clinic’s WhatsApp/phone number before leaving
- Advise them to send photos at 1 hour, 4 hours, and 24 hours post-application
- Intense tightness and darkening are normal with TCA — reassure the patient but emphasize: when in doubt, wash it off
- The patient should NOT be alarmed by the skin turning significantly darker over Days 1–2 — this is the expected TCA response before peeling begins
Post-Procedure Care Instructions
Provide these instructions to the patient (verbal + printed/WhatsApp). Compliance is critical with TCA peels — non-compliance significantly increases the risk of PIH and complications.
Day 0 (Day of Procedure)
- Leave the peel on for 4–6 hours as instructed (based on session number)
- Wash off thoroughly with lukewarm water
- Apply a thick recovery moisturizer or barrier cream immediately after
- No other actives, no makeup for the rest of the day
- Sleep on a clean pillowcase — expect some product transfer
Days 1–2 (Tightness & Darkening Phase)
- Skin will feel extremely tight, dry, and will appear significantly darker — the skin may take on a leathery, bronzed appearance. This is expected with TCA
- Cleanse with a gentle, soap-free cleanser once daily (avoid rubbing)
- Apply a thick, bland moisturizer or barrier cream 4–6 times daily — keep the skin heavily moisturized
- Apply SPF 50+ broad-spectrum sunscreen every 2 hours if going outdoors — UV exposure during this phase can cause permanent PIH
- Absolutely no direct sun exposure, saunas, steam rooms, hot showers, or strenuous exercise
- Do NOT pick, pull, scratch, or scrub under any circumstances
Days 3–5 (Heavy Peeling Begins)
- Heavy peeling and shedding will begin — this is significantly more intense than with AHA peels
- Do NOT pick, pull, or force peeling skin off — premature removal risks scarring and PIH
- Continue thick moisturizer 4–6 times daily + SPF 50+
- Large sheets of skin may come off — this is normal for TCA at this depth
- The skin underneath will appear pink and raw — this is expected
Days 5–8 (Active Peeling Continues)
- Peeling continues, gradually diminishing
- Continue moisturizer + SPF 50+ regimen
- New skin will be sensitive and pink — protect it from all irritants
- Avoid all actives, exfoliants, retinoids, and fragranced products
Days 8–10 (Recovery Phase)
- Peeling should be mostly complete by Day 8–10
- Continue moisturizer + SPF 50+
- May resume very light mineral makeup once peeling is complete
- Continue avoiding harsh actives (retinoids, AHAs, BHAs) until Day 14
- Fresh skin will appear significantly lighter and more even
Days 10–28 (Between Sessions)
- Maintain strict sun protection (SPF 50+, physical avoidance, hats)
- Can resume normal skincare routine from Day 14 onwards
- Consider maintenance with brightening serums (niacinamide, vitamin C) between sessions
- Next session at Day 21–28 — do not schedule earlier
Expected Peeling Timeline
Treatment Program
| Parameter | Recommendation |
|---|---|
| Total Sessions | 3–4 sessions (assess at each session) |
| Session Interval | 21–28 days between sessions (3–4 weeks) |
| Contact Time | 4–6 hours (self-neutralizing with frost monitoring) |
| Downtime | 5–10 days significant peeling per session |
| Full Program Duration | 3–4 months (3–4 sessions at 21–28 day intervals) |
| Product per Session | Approximately 3–4ml (full face) |
| Yield per Jar | ~7 full-face sessions from one 30ml jar |
| Maintenance | 1 session every 6–8 weeks, or step down to 580 protocol |
Progressive Protocol Guide
This protocol uses a progressive intensity approach. Increase application density and contact time gradually across sessions based on patient tolerance and frost response:
| Session | Contact Time | Approach |
|---|---|---|
| Session 1 | 4 hours | Conservative application, monitor frost closely, document frost level and patient response. This is the calibration session. |
| Session 2 | 4–5 hours | Increase application density if Session 1 was well tolerated with no PIH. 3–4 weeks after Session 1. |
| Session 3 | 5–6 hours | Full application if prior sessions tolerated. Assess cumulative clinical improvement. 3–4 weeks after Session 2. |
| Session 4 (if needed) | 6 hours | Maximum application and contact time. Only if substantial melasma remains. Do not exceed 6 hours. |
Expected Outcomes
- After Session 1: Significant peeling over 5–7 days. Initial lightening of superficial pigment. Skin texture noticeably smoother. This session calibrates the patient’s response.
- After Session 2: Deeper pigment deposits begin to lighten. Visible improvement in melasma patches. More even skin tone emerging.
- After Session 3–4: Substantial melasma reduction. Significantly more even complexion. Dermal pigment visibly diminished. Results comparable to traditional cosmelan/dermamelan protocols.
Set realistic expectations: Melasma is a chronic condition. While prodermic 612 can produce dramatic improvement, patients should understand that maintenance treatments and strict sun protection are essential for sustaining results. Melasma can recur with hormonal changes and UV exposure.
Full Ingredient List
Glycolic Acid, Lactic Acid, Trichloroacetic Acid (TCA), Arbutin, Kojic Acid, Hyaluronic Acid, Niacinamide, Vitamin C (Ascorbic Acid)

