|
clinical documentation
|
clinical documentation
555 PROCEDURE GUIDE
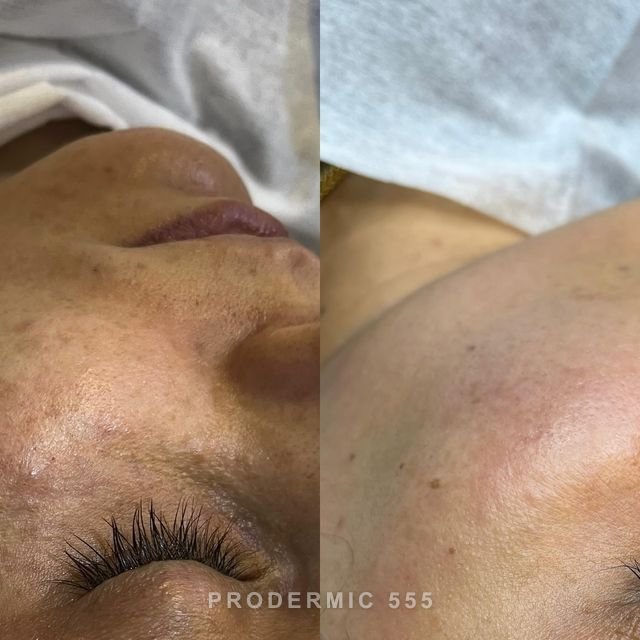
Depigment Protocol — Step-by-Step Clinical Application Guide for Practitioners
Product Overview

prodermic® 555
Depigment Protocol · 5-Agent Combo Peel
| Type | 5-Agent Combo Peel |
| Total Active | 5-agent combination |
| Size | 30ml jar |
| Yield | ~24 sessions per jar |
| Fitzpatrick | III–V |
| Peel Depth | Superficial |
Active Composition
Mechanism of Action
| Agent | Mechanism |
|---|---|
| Arginine | Amino acid and nitric oxide precursor — enhances microcirculation and active ingredient delivery to pigmented areas. Accelerates cellular repair in pigment-damaged skin. |
| Mandelic Acid | Lipophilic AHA with large molecular weight — slower, more even penetration avoiding patchy results common with glycolic-only systems. Broad-spectrum antibacterial. Works well across oily and dry zones. |
| Kojic Acid | Chelates copper ions required for enzymatic melanogenesis, providing sustained pigment-blocking action at the tyrosinase level. |
| Lactic Acid | Hydrating exfoliation with ceramide synthesis support. Strengthens skin barrier while promoting controlled desquamation of pigmented keratinocytes. |
| Glycolic Acid | Surface-level exfoliation — disrupts corneocyte cohesion to accelerate turnover of hyperpigmented superficial layers. |
Clinical Indications
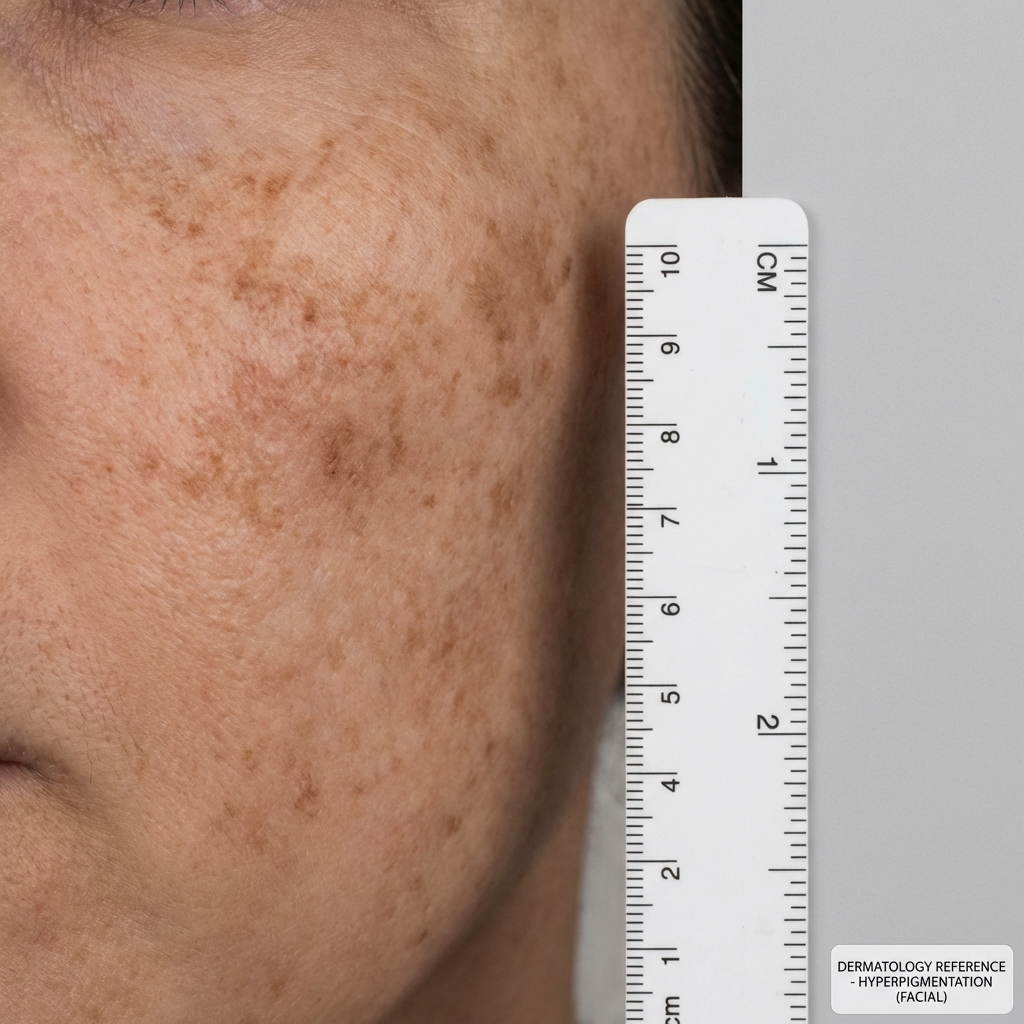
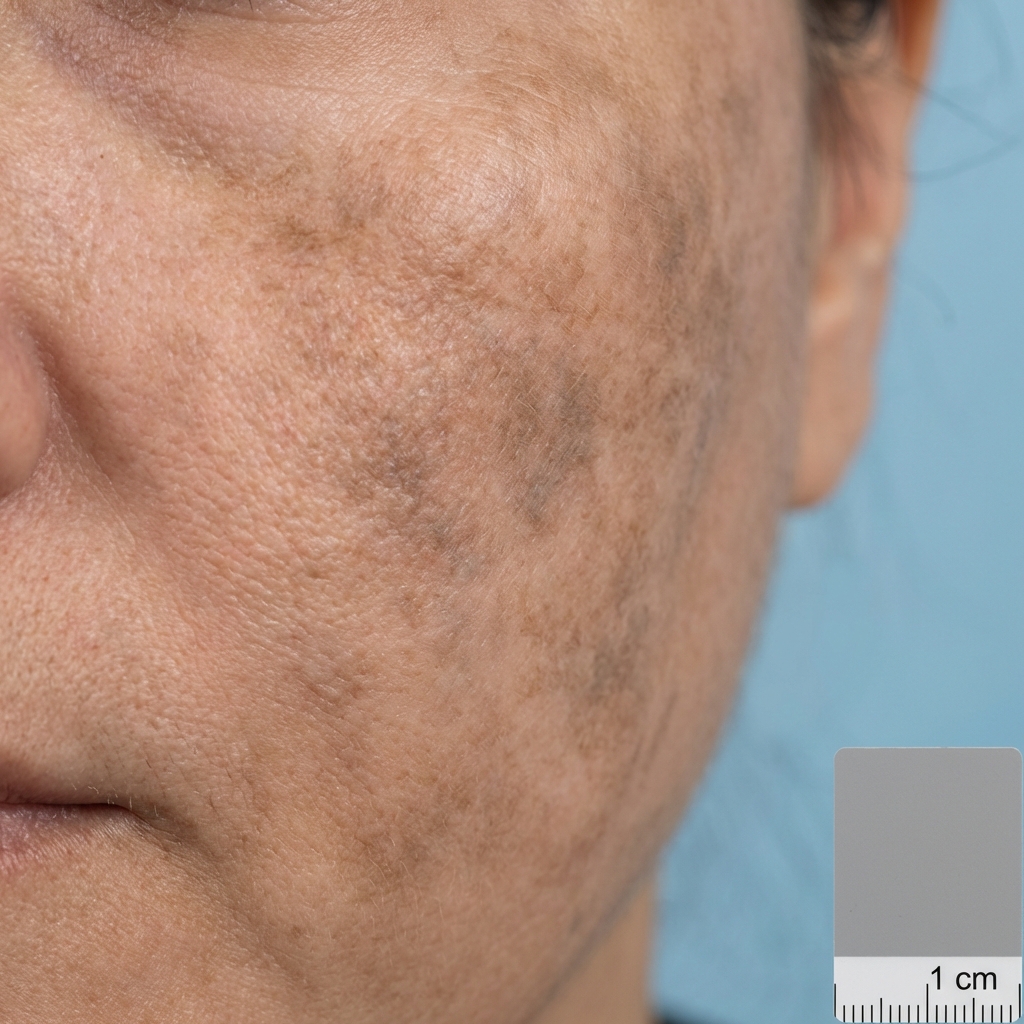
Patient Selection & Pre-Procedure
Ideal Patient Profile
- Stubborn post-inflammatory hyperpigmentation (PIH) unresponsive to standard glycolic/lactic protocols
- Patients needing cellular repair alongside depigmentation
- Deeper pigmentation that needs amino acid-enhanced delivery for better active penetration
- Fitzpatrick skin types III–V (formulated for darker skin tones)
- Patients with stubborn melasma requiring multi-mechanism depigmentation
Contraindications
DO NOT TREAT IF
- Active skin infections in the treatment area
- Compromised skin barrier (eczema, dermatitis flare)
- Pregnancy or breastfeeding (consult physician)
- Oral isotretinoin use within the last 6 months
- Topical tretinoin/retinoid use within 72 hours of session
- Open wounds, cuts, or abrasions in the treatment area
- Known allergy/sensitivity to any of the active ingredients (arginine, mandelic acid, kojic acid, lactic acid, glycolic acid)
Pre-Procedure Patient Preparation
- Skin Assessment: Evaluate skin type, pigmentation depth and distribution, melasma pattern, and prior peel/depigmentation history to determine suitability.
- Sun Exposure: Advise patient to avoid direct sun exposure for at least 1 week prior.
- Discontinue: Stop topical retinoids 72 hours before. Stop AHAs/BHAs 48 hours before.
- Informed Consent: Discuss expected outcomes, downtime (3–5 days of peeling/flaking), self-neutralizing nature (patient washes off at home), and possible side effects.
- Photography: Take standardized baseline photographs under consistent lighting. Use a Wood’s lamp if available to assess pigment depth.
Step-by-Step Procedure
KEY: SELF-NEUTRALIZING FORMULA
prodermic 555 is a self-neutralizing peel. It is applied in-clinic and the patient leaves with the peel on. The peel self-neutralizes over 3–5 hours. The patient washes it off at home after the contact period. No in-clinic neutralization is required.
- Forehead: Apply in horizontal strokes from center outward
- Cheeks: Apply from the nose outward toward the ears — focus on pigmented patches
- Nose: Apply evenly across the bridge and sides
- Chin: Apply in upward strokes
- Upper lip: Light, thin application (sensitive area, common melasma zone)
- Mild warmth + tingling: Normal — this is the expected initial response from the multi-acid formula
- Moderate stinging: Monitor for 2–3 more minutes; usually subsides as the peel begins self-neutralizing
- Severe burning or swelling: Remove product immediately with cold water
- Leave the peel on: The patient leaves with the peel on their face
- Contact time: Instruct the patient to leave it on for 3–5 hours
- Wash off at home: After 3–5 hours, rinse thoroughly with lukewarm water until all residue is removed
- Apply a soothing recovery moisturizer immediately after washing
- No other actives or makeup for the rest of the day
- Pat skin dry gently — do not rub
- Apply a thick, bland recovery moisturizer or balm generously
- Do not apply any actives, serums, or makeup until the next morning
- Sleep on a clean pillowcase
Early Removal & Adverse Reactions
INSTRUCT PATIENT TO WASH OFF IMMEDIATELY IF
- Severe burning or pain that does not subside or is escalating
- Excessive redness or swelling beyond mild warmth
- Hives, welts, or allergic reaction signs
- Blistering or skin breakdown
- Inability to tolerate the sensation
Emergency Removal Steps (At Home)
- Rinse the treatment area thoroughly with cold water until all product is removed
- Continue rinsing until all tingling/burning completely stops
- Apply a thick, bland moisturizer (no actives)
- Apply a cold compress for comfort if needed
- Contact the clinic immediately to report the reaction
- Document the reaction — reduce contact time or consider a milder protocol for the next session
PATIENT COMMUNICATION
- Ensure the patient has the clinic's WhatsApp/phone number before leaving
- Advise them to send a photo if they notice anything unusual
- Mild warmth and tightness are normal — reassure the patient
- If in doubt, instruct them to wash it off early — better safe than sorry
Post-Procedure Care Instructions
Provide these instructions to the patient (verbal + printed/WhatsApp):
Day 0 (Day of Procedure)
- Leave the peel on for 3–5 hours as instructed
- Wash off thoroughly with lukewarm water
- Apply recovery moisturizer immediately after
- No other actives, no makeup for the rest of the day
- Sleep on a clean pillowcase
Days 1–2 (Tightness Phase)
- Skin will feel tight, dry, and may appear slightly darkened — this is expected
- Cleanse with a gentle, soap-free cleanser twice daily
- Apply a thick, bland moisturizer or recovery balm 3–4 times daily
- Apply SPF 50+ broad-spectrum sunscreen every 2–3 hours if going outdoors
- Avoid direct sun exposure, saunas, steam rooms, strenuous exercise
- Do NOT pick, pull, or scrub — even if skin feels rough
Days 3–5 (Peeling Phase)
- Visible flaking and peeling typically begins around Day 3
- Do NOT pick, pull, or force peeling skin off — let it shed naturally
- Continue moisturizer 3–4 times daily + SPF 50+
- Pigmented areas may peel more visibly — this is a positive sign of the depigmentation process working
Days 5–7 (Recovery Phase)
- Peeling should be mostly complete by Day 5–7
- Continue moisturizer + SPF 50+
- May resume light makeup once peeling is complete
- Continue avoiding harsh actives (retinoids, AHAs, BHAs) until Day 7
Days 7–14 (Between Sessions)
- Maintain strict sun protection (SPF 50+, physical avoidance) — critical for depigmentation outcomes
- Can resume normal skincare routine
- Consider prescribing maintenance depigmenting serums between sessions for continued pigment suppression
Expected Peeling Timeline
Treatment Program
| Parameter | Recommendation |
|---|---|
| Total Sessions | 4–6 sessions (assess at session 3) |
| Session Interval | 14 days between sessions |
| Contact Time | 3–5 hours (self-neutralizing, washed off at home) |
| Downtime | 3–5 days peeling/flaking per session |
| Full Program Duration | 2–3 months (4–6 sessions at 14-day intervals) |
| Product per Session | Approximately 1–1.25ml (full face) |
| Yield per Jar | ~24 full-face sessions from one 30ml jar |
| Maintenance | 1 session every 4–6 weeks after initial program, or as needed |
Progressive Protocol Guide
Adjust contact time progressively across sessions based on patient tolerance:
| Session | Contact Time | Notes |
|---|---|---|
| Session 1 | 3 hours | Conservative first exposure. Assess skin response and tolerance the next day. |
| Session 2 | 3–4 hours | Increase if Session 1 was well tolerated with no adverse effects. |
| Session 3 | 4 hours | Assess clinical depigmentation results so far. Adjust protocol if needed. |
| Session 4+ | 4–5 hours | Maximum contact time. Do not exceed 5 hours. |
Expected Outcomes
- After Session 1: Visible lightening of superficial pigmentation. Skin appears fresher with improved luminosity.
- After Session 2–3: Noticeable improvement in post-inflammatory hyperpigmentation patches. More even skin tone emerging.
- After Session 4–6: Significant reduction in deep pigmentation and melasma. Clearer, more uniform complexion with restored radiance.
Set realistic expectations: Deep, longstanding pigmentation (melasma, dermal PIH) may require the full 6-session program and ongoing maintenance. Sun protection compliance is critical — any UV exposure between sessions can reverse depigmentation progress.
Full Ingredient List
Arginine, Mandelic Acid, Kojic Acid, Lactic Acid, Glycolic Acid, Liquorice Extract, Tea Tree Extract